Introducing New Next-Generation 8-Station Tablet Dissolution Testing Instrument PTWS 830
We are pleased to announce the upcoming launch of its next-generation PTWS tablet dissolution testing instruments. They are available for order now and will begin shipping in the first quarter of 2026. Designed with a modern graphical user interface, advanced user management features, and a fully integrated audit trail, the new systems set a new benchmark in usability and compliance.
Available Models
Three new models of tablet dissolution testing instruments are available to order now and will begin shipping in the first quarter of 2026:
- PTWS 830: Eight-station dissolution tester
- PTWS 1230: Twelve-station dissolution tester
- PTWS D630: “Dual drive” six-plus-six station dissolution tester
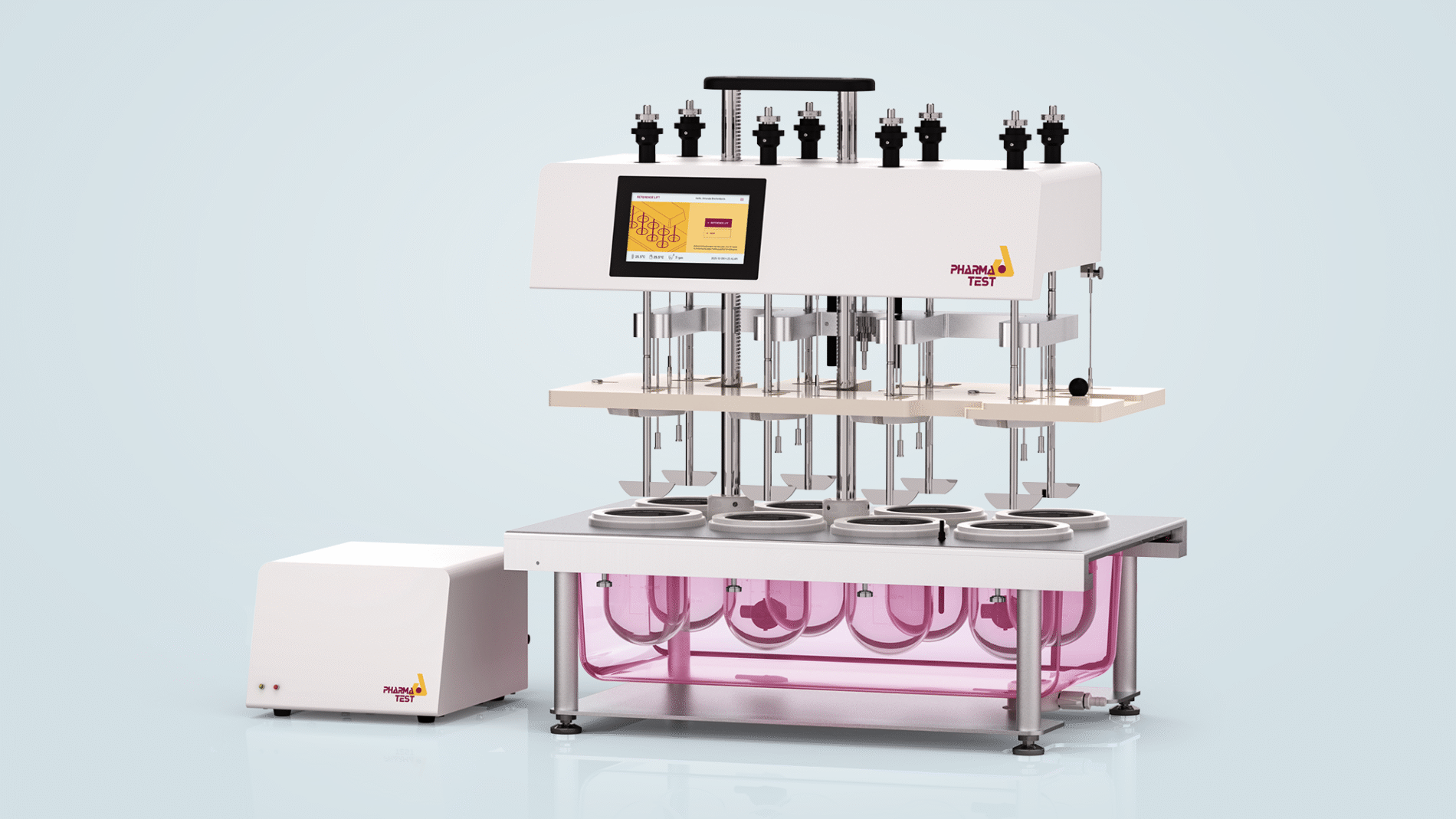
PTWS 830 Instrument Features
PTWS 830 is a compact instrument equipped with 8 testing positions. It arrives fully configured and ready for immediate use. Each package includes a complete set of USP vessels, stirrer shafts, and USP apparatus 2 paddles, ensuring compliance with regulatory standards and facilitating efficient laboratory workflows. The instrument still uses the Pharma Test MonoShaft design and supports staggered stirrer start for manual operation.